|
So, whichever route you took, of the five options given, the one that was an alkali metal was cesium. The electropositive element or group releases its electrons i.e. Both metals and nonmetals are classified as electropositive and electronegative. Be careful when looking at the periodic table and make sure the name as well as the symbol matches up. The elements in the periodic table are divided into two parts: alkali metal and alkaline earth metals. They all have one electron in their outer shell, which results in weak metallic bonding. They include lithium, sodium, potassium, etc. Alkaline earth metals are elements found in group IIA that share properties characteristic of metals but form hydroxides when reacted with water and ions. This is the first column of the periodic table. The alkali metals are all members of group 1 on the periodic table, minus hydrogen. Three of the options were elements with symbols that began with C. In chemistry, it refers to a base that can dissolve in water. Therefore, it’s not an alkali metal, and calcium is an incorrect answer.Īnother way of doing this would be to list all the alkali metals, lithium, sodium, potassium, rubidium, cesium, and francium, and finding which one overlapped with the options. Calcium sits in group two of the periodic table. 
A transition metal is defined by IUPAC as an element whose atom has a. Lanthanum is not in group one, so it’s not a correct answer. The alkali metals form Group I of the periodic table. The alkaline earth metals can be found in. These metals are highly reactive and form ionic compounds (when a nonmetal and a metal come together) as well as many other compounds. Alkaline earth metals are sometimes called group 2 elements because they are located in the second group or column of the periodic table of elements. Various properties of the group 1 elements are summarized in Table 20.4. Lanthanum has the symbol La and is the first element in the lanthanides. The Alkali metals are comprised of group 1 of the periodic table and consist of Lithium, Sodium, Rubidium, Cesium, and Francium. So, we can be pretty confident that cesium is the correct answer.īut just to be safe, I’m going to go through the other two options. Cesium is a base with the OH− group that produces alkaline solutions. The only element in the first column that is not usually considered an alkali metal is hydrogen. Going back to what we’ve said about alkali metals forming alkaline solutions upon reaction with water, cesium reacts with water to produce a solution of cesium hydroxide and hydrogen. They are all in the first column of the periodic table. Since cesium is a metal in group one, it is an alkali metal and is, therefore, the correct answer. What about cesium with symbol Cs? Cesium is second from the bottom of group one. So, cerium is not in group one, and cerium is an incorrect answer. Meanwhile, cerium has the symbol Ce and sits in the second position in the lanthanides. Since radium is not in group one, it is not a correct answer. Radium has the symbol Ra and sits at the bottom of group two. Now that we have the periodic table, we can look for each element and see which one lies in group one. And then as we go from left to right on the periodic table, as we go from Alkali Metal to Noble Gases we see that the ionization energy increases. So, it takes very low energy to remove that electron. You remove an electron, it gets to a stable state. But hydrogen is not a metal and, therefore, is not an alkali metal. Lithium, as we said, this is an Alkali metal. Hydrogen is the first member of group one. Group one is the first column of the periodic table and can be found here. However, the modern definition relies on their presence in group one. 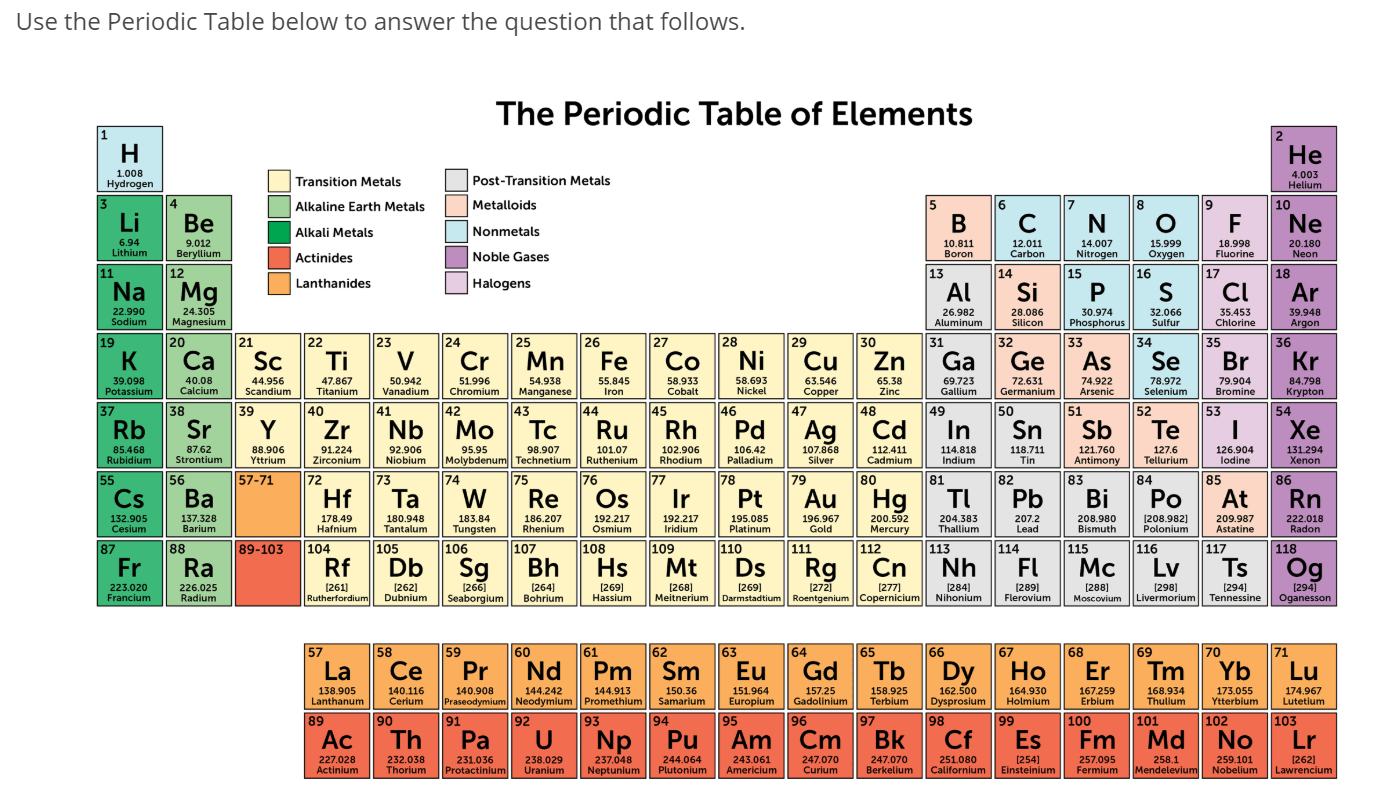
Alkali metals get their name from the fact they form alkaline solutions upon reaction with water. An alkali metal is a member of group one on the periodic table. The first thing we need to do is define an alkali metal. Alkali metals are any of the elements found in Group IA of the periodic table (the first column). Learn more about the occurrence and uses of lithium. The metal itselfwhich is soft, white, and lustrousand several of its alloys and compounds are produced on an industrial scale. It is the energy released (per mole of X) when this change happens. The first electron affinity is the energy released when 1 mole of gaseous atoms each acquire an electron to form 1 mole of gaseous -1 ions. Melting and boiling points of the alkali metals and selected reference substances.Which of the following is an alkali metal? A) Radium. Lithium, chemical element of Group 1 (Ia) in the periodic table, the alkali metal group, lightest of the solid elements. Electron affinities are the negative ion equivalent, and their use is almost always confined to elements in groups 16 and 17 of the Periodic Table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
